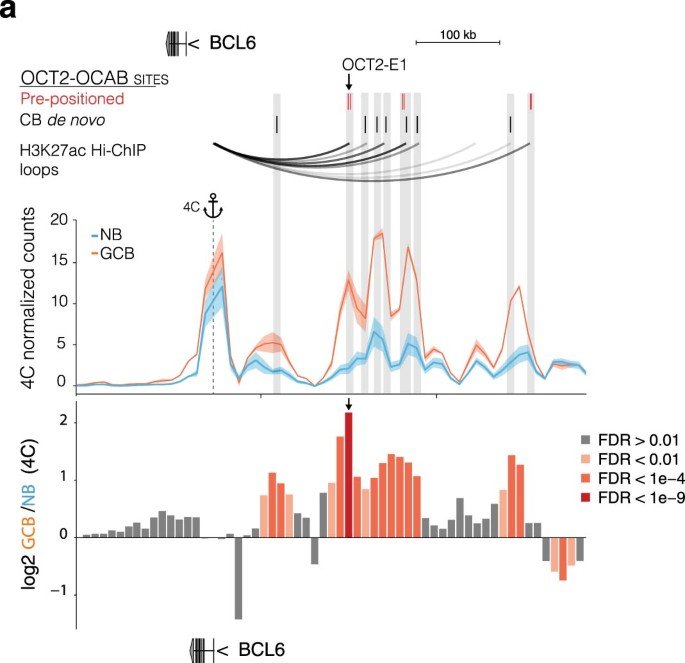
OCT2 pre-positioning facilitates cell fate transition and chromatin architecture changes in humoral immunity
During the germinal center (GC) reaction, B cells undergo profound transcriptional, epigenetic and genomic architectural changes. How such changes are established remains unknown. Mapping chromatin accessibility during the humoral immune response, we show that OCT2 was the dominant transcription factor linked to differential accessibility of GC regulatory elements. Silent chromatin regions destined to become GC-specific super-enhancers (SEs) contained pre-positioned OCT2-binding sites in naive B cells (NBs). These preloaded SE 'seeds' featured spatial clustering of regulatory elements enriched in OCT2 DNA-binding motifs that became heavily loaded with OCT2 and its GC-specific coactivator OCAB in GC B cells (GCBs). SEs with high abundance of pre-positioned OCT2 binding preferentially formed long-range chromatin contacts in GCs, to support expression of GC-specifying factors. Gain in accessibility and architectural interactivity of these regions were dependent on recruitment of OCAB. Pre-positioning key regulators at SEs may represent a broadly used strategy for facilitating rapid cell fate transitions.
Journal: Nature Immunology PMID: 34556886 DOI: 10.1038/s41590-021-01025-w

Histone H1 loss drives lymphoma by disrupting 3D chromatin architecture
Based on gene expression profiles, diffuse large B cell lymphoma (DLBCL) is sub-divided into germinal center B cell-like (GCB) and activated B cell-like (ABC) DLBCL. Two of the most common genomic aberrations in ABC-DLBCL are mutations in MYD88, as well as BCL2 copy number gains. Here, we employ immune phenotyping, RNA-Seq and whole exome sequencing to characterize a Myd88 and Bcl2-driven mouse model of ABC-DLBCL. We show that this model resembles features of human ABC-DLBCL. We further demonstrate an actionable dependence of our murine ABC-DLBCL model on BCL2. This BCL2 dependence was also detectable in human ABC-DLBCL cell lines. Moreover, human ABC-DLBCLs displayed increased PD-L1 expression, compared to GCB-DLBCL. In vivo experiments in our ABC-DLBCL model showed that combined venetoclax and RMP1-14 significantly increased the overall survival of lymphoma bearing animals, indicating that this combination may be a viable option for selected human ABC-DLBCL cases harboring MYD88 and BCL2 aberrations.
Journal: Nature PMID: 33299181 DOI: 10.1038/s41586-020-3017-y
